  
For a substance decaying exponentially, the amount of time it takes for the amount of the substance to diminish by half. Since the half life of Carbon 14 is 5730 years, this means that after 5730 years there will only be 5 micrograms of Carbon 14 left in the preserved plant: f(. However, if you must learn about these in school, then this is the place to. & pre-calculus : calculus: advanced topics: probability & statistics: real world applications: multimedia entries: about mathwords : website feedback : Half-Life. The following equation gives the quantitative relationship between the original number of nuclei present at time zero (N_0\) and the number (\(N\)) at a later time \(t\). Actually, you dont need to know about radioactive decay constants,, 'k', etc to do half-life calculations. It is also applicable to the decay of excited states in atoms and nuclei. 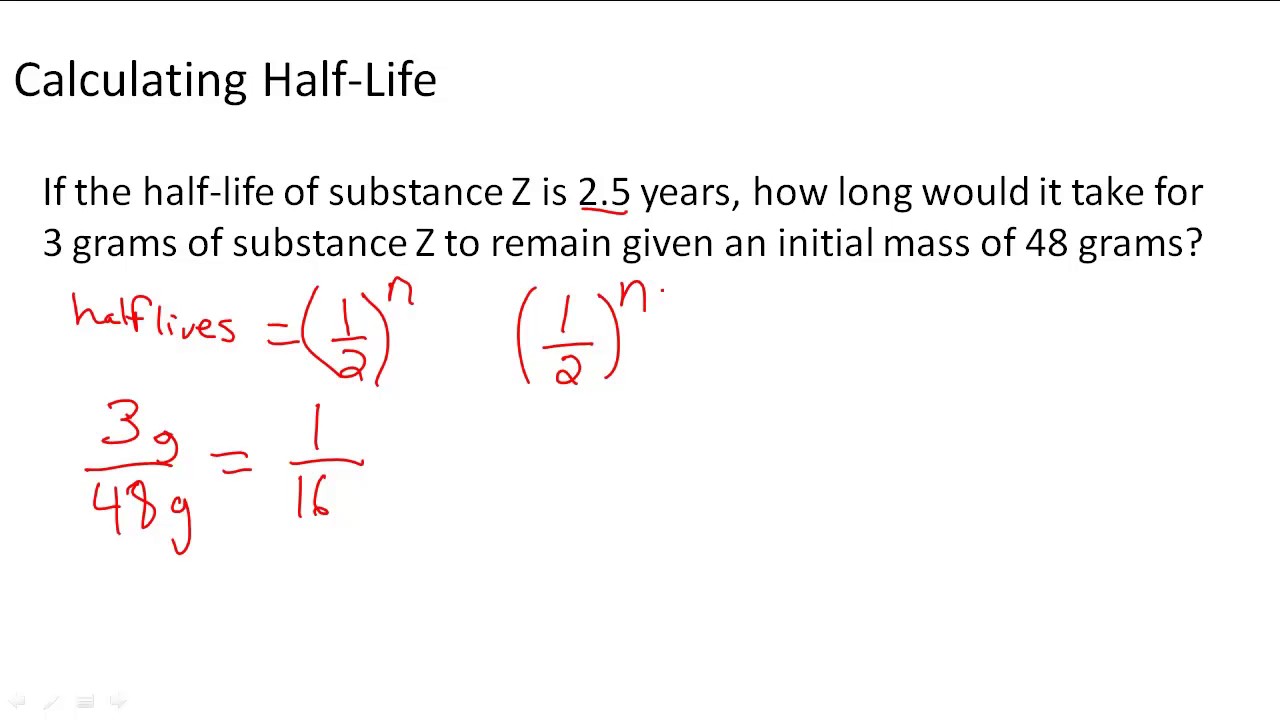
We use half-life in applications involving radioactive isotopes. Finally, well use the first-order half-life equation to calculate the half-life of the reaction. Well also calculate the amount of time it takes for the concentration to decrease to a certain value. The dependence of the order of fractional derivative. The concept of half-life is applicable to other subatomic particles, as will be discussed in Particle Physics. We may use the exponential decay model when we are calculating half-life, or the time it takes for a substance to exponentially decay to half of its original quantity. In this video, well use the first-order integrated rate law to calculate the concentration of a reactant after a given amount of time. Consequently, the theoretical half-life values have been obtained in consistent with the experimental data. For instance, half life of plutonium-239 is 24110 years, half life of caesium-135 is 2.3 milliards years, halves living of radium-224 is only a few days. t1 2 t 1 2 0.693/ 0.693/0.84 0.825 Therefore, the half life of the substance is 0.8 seconds. For example, this feature can representing adenine highly decay of certain quantity regarding plutonium-239 and describes the amount of plutonium-239 left after a time period t. This stuff just wont go away Hey, did you. Solution: Given decay constant 0.84 The half life formula can be used to find the half life of the substance. RADIOACTIVE DECAY: Ever heard of Plutonium Half-life means that, if you have 100 pounds of Plutonium-239. Nuclides with the shortest half-lives are those for which the nuclear forces are least attractive, an indication of the extent to which the nuclear force can depend on the particular combination of neutrons and protons. Half-life is the time required for the amount of something to fall to half its initial value. Example 1: The decay constant of a substance is 0.84 s-1. During the next 3 years, 12.5 grams would remain and so on. AP Calculus: Exponential Growth/Decay (Half Life) Christopher Manker. For example, if the half-life of a 50.0 gram sample is 3 years, then in 3 years only 25 grams would remain. Rewrite the function in terms of half-life.\)y for the least unstable, or about 46 orders of magnitude. A radioactive half-life refers to the amount of time it takes for half of the original isotope to decay. Youd wait a reasonable about of time (a year), use the natural log to find the continuous rate over that period, and work out the half life. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
